Epoxidation of Geraniol
Epoxidation of Geraniol: A Quest to Determine the Enantioselectivity of Two Epoxidation Procedures
By: Ryan DeAngelis, Andre Mound
Abstract
The purpose of this experiment was to compare the enantioselectivity of the meta-chloroperoxybenzoic acid epoxidation procedure and the Sharpless Asymmetric Epoxidation. The enantioselectivity of each procedure in the epoxidation of geraniol was investigated via chiral shift 1H-NMR spectroscopy. The results indicated that the mCPBA epoxidation procedure did not exhibit enantioselectivity, whereas the Sharpless Asymmetric Epoxidation demonstrated enantioselectivity for the (2S, 3S)-epoxy geraniol. The mCPBA epoxidation showcased enantiomeric excess values of 1.6% and 4%, whereas the Sharpless Asymmetric Epoxidation displayed enantiomeric excess values of 83.5% and 76.2%. These results are in agreement with the literature, and ultimately serve as an example of the asymmetric power of the Sharpless Asymmetric Epoxidation.1
Introduction
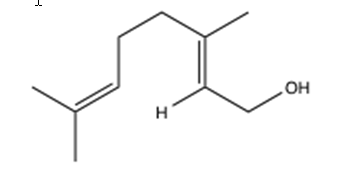
The purpose of this experiment is to compare two epoxidation procedures—a standard epoxidation procedure that uses meta-chloroperoxybenzoic acid (mCPBA) and the Sharpless Asymmetric Epoxidation reaction.1,2 The enantioselectivity of each epoxidation reaction will be examined via chiral lanthanide shift reagents and 1H-NMR spectroscopy. Figure 1 below shows the structure of the starting material, geraniol.
Figure 1: Structure of the starting material, geraniol.
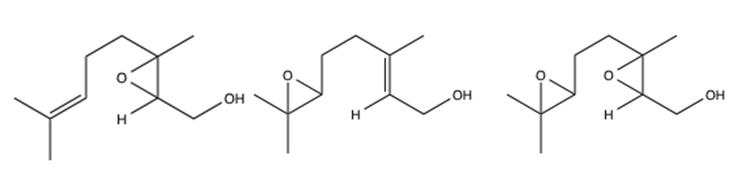
An epoxide is a cyclic ether with three ring atoms.3 There are multiple synthetic methods for creating epoxides, but the focus of this experiment is the epoxidation of an allylic alcohol. Theoretically, both double bonds in geraniol are capable of reacting with the epoxidation reagents. Thus, there are multiple epoxide products that can form.2 The possible epoxidation products, ignoring stereochemistry, are shown in Figure 2 below.
Figure 2: Structure of 2,3-epoxygeraniol, 6,7-epoxygeraniol, and 2,3,6,7-diepoxygeraniol, respectively. Stereochemistry is ignored.
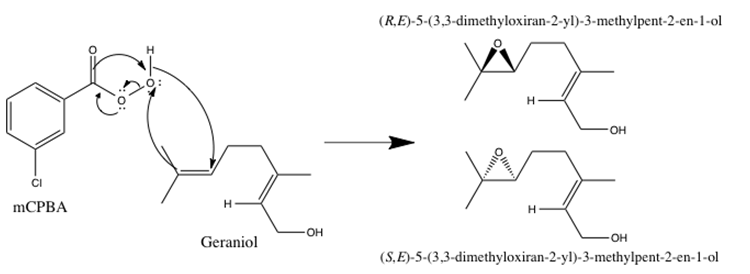
Understanding the mechanism of each epoxidation procedure is crucial in order to predict which of the possible products can form. The mCPBA epoxidation procedure utilizes the peroxycarboxylic acid functional group of the meta-chloroperoxybenzoic acid to deliver one of the peroxide oxygens across the double bond.3 A detailed mechanism of the mCPBA epoxidation of geraniol at the 6,7 alkene position is shown in Figure 3 below.
Figure 3: Mechanism for the mCPBA epoxidation of geraniol to yield both isomers of 6,7-epoxygeraniol (the IUPAC names are shown in the figure above). The monoepoxidation can also occur at the 2,3 position, and a diepoxidation can occur at both the 2,3 and 6,7 position.
The carbon atoms involved in the double bond are sp2 hybridized, and therefore, the bond is planar. Thus, the peroxide can attack from either face of the alkene, forming racemic mixtures of both the monoepoxide products as well as the diepoxide product.1,3 Thus, the hypothesis for the mCPBA epoxidation procedure is that there will be minimal enantioselectivity for both the monoepoxide and diepoxide products.
It is also important to discuss the specific role of each reagent used in the mCPBA procedure. As discussed above, mCPBA contains a peroxycarboxylic acid functional group, which donates a single oxygen atom to the alkene to form the epoxide. However, this mechanism generates excess peroxides throughout the reaction. Sodium thiosulfate is utilized to reduce the excess peroxides and minimize the chance of an explosion while concentrating the product to dryness. Sodium bicarbonate is also introduced to serve as a buffer by removing the acidic byproduct.1,2,3
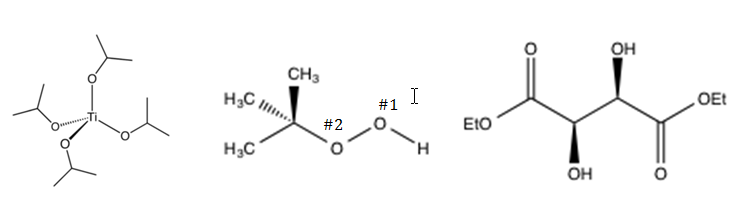
The Sharpless Asymmetric Epoxidation procedure is recognized as a powerful asymmetric synthetic method.1 Asymmetry in the epoxidation of the allylic alcohol is induced by utilizing a catalyst, titanium (IV) tetraisopropoxide (Ti(OiPr)4), a peroxide, tert-butylhydroperoxide (TBHP), and a dialkyl tartrate, L-(+)-diethyltartrate (L-(+)-DET). The structure of each reagent involved in the Sharpless Asymmetric Epoxidation procedure is shown in Figure 4 below.
Figure 4: Structures of Ti(OiPr)4, TBHP, and L(+)-DET, respectively.
The Sharpless Asymmetric Epoxidation mechanism is a catalytic cycle.4 Titanium (IV) tetraisopropoxide forms a complex with the diethyl tartrate and the allylic alcohol. Due to the oxygen of the allylic alcohol coordinating with the titanium, the epoxidation occurs at the 2, 3 alkene position.4 Additionally, the coordination of the Ti(OiPr)4 and DET creates a chiral environment for the reaction. Oxygen #1 of the TBHP (see “Figure 4” for labeling) nucleophilically attacks the titanium atom of the Ti(OiPr)4. The titanium atom then undergoes an intramolecular nucleophilic attack by Oxygen #2 of the TBHP. This induces Oxygen #1 to give its electrons to the neighboring positively charged Oxygen #2. Simultaneously, Oxygen #1 interacts with the pi electrons of the alkene to form the epoxide. Oxygen #1 is then cleaved from the titanium to yield the epoxy alcohol as another TBHP and allylic alcohol molecule bind to the Ti(OiPr)4 to continue the catalytic cycle.4
In order to development a deeper understanding of the Sharpless Asymmetric Epoxidation mechanism, it is important to understand the particular role of each reagent. Ti(OiPr)4 coordinates with diethyl tartrate to create a chiral environment for the reaction. The oxygen of allylic alcohol coordinates to this complex to facilitate the epoxidation of the 2,3 alkene position. TBHP is the oxidizing agent in the reaction; it delivers the oxygen atom across the alkene to form the epoxide. Similar to the mCPBA epoxidation procedure, sodium thiosulfate is used to reduce the excess peroxides created by the TBHP. The diethyl tartrate forms the chiral environment, and facilitates the stereoselective delivery of Oxygen #1 to the alkene.4
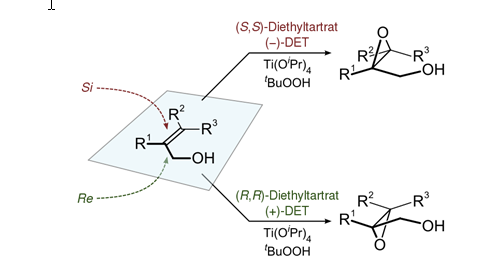
It is particularly important to discuss the specific isomer of diethyl tartrate utilized in this epoxidation procedure. There are two forms of diethyl tartrate: L-(+)-DET and D-(-)-DET. The L isomer facilitates the delivery of Oxygen #1 to the Re face of the allylic alcohol, whereas the D isomer facilitates the delivery of Oxygen #1 to the Si face of the allylic alcohol.5 Figure 5 shows the Re and Si attack of an allylic alcohol.
Figure 5: Re and Si attack of an allylic alcohol using D-(-)-DET and L-(+)-DET. The allylic alcohol in this experiment is geraniol (see “Figure 1”).
Being that each of the DET isomers induce the formation of a specific stereoisomer, and the allylic alcohol coordinating to the titanium facilitates the epoxidation of the 2, 3 alkene position, it is said that the Sharpless Asymmetric Epoxidation is both stereoselective and regioselective.1,4 Thus, the hypothesis for the Sharpless Asymmetric Epoxidation procedure is that it will demonstrate enantioselectivity and the oxygen will add to the Re face of the allylic alcohol at the 2,3 alkene position.
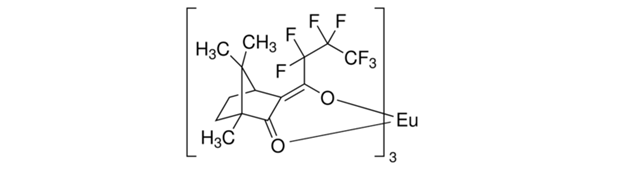
In order to determine the enantioselectivity of each epoxidation procedure, a chiral lanthanide shift reagent and 1H-NMR spectroscopy are utilized.1,2 The chiral lanthanide shift reagent, Europium tris[3(heptafluoropropylhydroxymethylene)-(+)- camphorate] (Eu(hfpc)3), is shown in Figure 6 below.
Figure 6: Structure of the chiral lanthanide shift reagent utilized in this experiment, Eu(hfpc)3.
Enantiomers are not distinguishable in an achiral environment, and therefore, cannot be differentiated between via 1H-NMR spectroscopy. Introducing Eu(hfpc)3 to the system allows the enantioselectivity of each epoxidation procedure to be determined.6 The chiral lanthanide shift reagent is a Lewis Acid, and thus, able to accept electrons.7 Theoretically, the alcohol oxygen could serve as the Lewis Base, but the alcohol oxygen is not significantly electronically different than the epoxide oxygen. Therefore, the epoxy geraniol is further functionalized to the epoxy acetate by introducing acetic anhydride to the system. The Eu3+ chemoselectively coordinates to the carbonyl oxygen of the acetate due to its high electron density, and ultimately, forms a diastereomeric interaction that allows the enantiomers to be distinguished via 1H-NMR.7 This coordination induces a spreading of the chemical shifts.7 The signal generated from the enantiomeric protons at position 1, originally at 3.0 ppm, shifts downfield and splits into two separate peaks. The integration values of each peak represent the relative amount of each specific enantiomer.1,2 Thus, by converting the epoxy geraniol to the epoxy acetate and performing the chiral 1H-NMR shift analysis, the enantioselectivity of each epoxidation procedure can be determined.
Column chromatography is crucial for isolation and purification of the desired products in this experiment. The crude epoxy geraniol product theoretically contains a mixture of the 2,3-epoxy geraniol, 6,7-epoxy geraniol, and 2,3,6,7-diepoxy geraniol. The column is packed with a slurry of silica gel dissolved in a specific eluent and the crude product is loaded onto the column. Being that silica is a particularly polar molecule, the different epoxide products present in the crude product are separated based on their polarity. The more polar compounds interact more with the polar silica gel on the column than the less polar compounds; therefore, the more polar compounds elute slower. Additionally, the polarity of the eluent is adjusted to assist in the separation of the product. The differing polarities of each epoxide product allow for each product to be separated and purified for the acetate formation step. Thin-layer chromatography is utilized to determine which column fractions contain the desired product, and the fractions containing the desired epoxide product are then concentrated to dryness to yield the purified epoxide product. After acetate formation, column chromatography is then utilized again to isolate the desired acetate product for the chiral shift 1H-NMR experiment.
In terms of a “real-world” application, the stereoselective and regioselective properties of the Sharpless Asymmetric Epoxidation are particularly useful in the enantioselective synthesis of various compounds. For example, the epoxide product is fairly reactive with a wide range of nucleophiles, and thus, can be treated with OH– and washed with dilute acid to yield a trans chiral vicinal diol.3 Chiral vicinal diols are commonly found in pharmaceutical drugs. The asymmetric power of the Sharpless Asymmetric Epoxidation allows for the stereoselective and regioselective synthesis of these chiral vicinal diols in high yield.
Experimental Procedure—Sharpless Epoxidation
The experimental procedure was very similar to the one outlined in the laboratory handout, with some minor modifications.2
L-(+)-Diethyltartrate (671 mg, 3.25 mmol, 1 equiv) was weighed directly into a dry 25 mL round-bottom flask equipped with a magnetic stir bar. Titanium tetraisopropoxide (960 mL, 3.24 mmol, 1 equiv) and dry dichloromethane (CH2Cl2, 10 mL) were then added to the flask. A septum was then attached to the flask and a syringe with a nitrogen balloon attached was inserted into the septum. The reaction mixture was stirred for five min. at -23O C in a salt and dry ice bath. Geraniol (510 mg, 3.31 mmol, 1.02 equiv) was mixed with dichloromethane (1 mL) and added to the flask via syringe. Tert-butylhydroperoxide (1.2 mL, 6.60 mmol, 2.04 equiv) was first poured into a small vial and then transferred to the flask via syringe. The reaction mixture was then stirred for 45 min. at -23O C, and an Infrared (IR) spectrum of geraniol was acquired while stirring. After stirring for 45 min., the flask was capped and stored in a -18O C freezer until the following week.
One week later, the flask was removed from the freezer and immediately placed in a -23O C acetone and dry ice bath. 10% tartaric acid (8.3 mL) was added to the flask and the reaction mixture was stirred for 30 min. at -23O C. The acetone and dry ice bath was then removed and the mixture was stirred at room temperature for 45 min. The mixture was transferred to a separatory-addition funnel, and an emulsion formed. Brine was added to the mixture, the mixture was transferred to a 25 mL round-bottom flask, and was placed in a -18O C freezer until the following week.
The following week, the reaction mixture was removed from the freezer and the aqueous and CH2Cl2 layers were clearly identifiable. The CH2Cl2 layer was separated from the top yellow aqueous layer via a separatory-addition funnel. The CH2Cl2 was then washed twice with an equal volume of sodium thiosulfate in order to reduce any excess peroxides. The organic layer was then washed twice with equal volumes of water, dried over anhydrous sodium sulfate (Na2SO4), and concentrated to dryness to yield a cloudy colorless oil. A mass of the crude epoxide product was obtained.
An 11 mm inner diameter by 300 mm long column was packed with a slurry of 200-400 mesh silica gel in 70% petroleum ether (PE)/30% ethyl acetate (EA). The crude epoxide was loaded onto the column via glass pipet and solvent was forced through the column to collect fifteen 5 mL fractions. Thin-layer chromatography (TLC) was utilized to check the fractions for the presence of the desired 2,3 epoxy geraniol product (silica gel on plastic backing, 70% PE/30% EA, potassium permanganate (KMnO4) dip used to visualize). Fractions 5-7 were combined and concentrated to dryness to yield a clear colorless oil. A mass of the desired epoxide product was obtained.
The purified 2,3-epoxygeraniol (229 mg, 1.35 mmol, 5 equiv) was transferred to a 25 mL round-bottom flask and dissolved in CH2Cl2 (10 mL). Dimethylaminopyridine (DMAP, 43 mg, 0.35 mmol, 1 equiv) and a magnetic stir bar were added to the flask. The flask was equipped with a rubber septum. Triethylamine (0.5 mL, 3.50 mmol, 10 equiv) was added to the flask and the reaction mixture was stirred for 30 min. at 0O C. While stirring, acetic anhydride (420 mL, 4.45 mmol, 12.71 equiv) was added dropwise via syringe. TLC was utilized to monitor the progress of the reaction (silica gel on plastic backing, 100% CH2Cl2, KMnO4 dip used to visualize). After stirring for 30 min., the reaction mixture was washed with an equal volume of H2O. The CH2Cl2 layer was then quickly washed with cold hydrochloric acid (HCl, 10 mL, 1 M), dried over Na2SO4, and concentrated to dryness to yield a yellow oil. A mass of the crude acetate product was obtained.
An 11 mm inner diameter by 300 mm long column was packed with a slurry of 200-400 mesh silica gel in 95% PE/5% EA. The crude acetate was loaded onto the column via glass pipet and solvent was forced through the column to collect fifteen 5 mL fractions. Thin-layer chromatography (TLC) was utilized to check the fractions for the presence of the desired acetate product (silica gel on plastic backing, 95% PE/5% EA, KMnO4 dip used to visualize). Due to the inability to visualize the epoxy acetate product on the TLC plate, TLC analysis was performed again with a different solvent system (silica gel on plastic backing, 70% PE/30% EA, KMnO4 dip used to visualize). Fractions 6-10 were combined and concentrated to dryness to yield a clear colorless oil. A mass of the epoxy acetate product was obtained. The epoxy acetate product was further concentrated to dryness and the mass was obtained again. This was repeated until the mass remained constant, indicating all solvent had been removed.
1H-NMR was acquired of the purified epoxy acetate product (used deuterated chloroform as solvent). The chiral shift reagent was then prepared by weighing Eu(hfpc)3 (35.0 mg) into a small, clean, and dry vial. Deuterated chloroform (CDCl3, 600 mL) was added, and the mixture was removed from the vial using a 1 mL plastic syringe. Another 1 mL plastic syringe was fitted with a plastic filter tip, and the mixture was forced through the filter tip with gentle pressure. The filtered solution was collected in a small, clean, and dry vial. The vial was capped immediately and labeled “Chiral Shift Reagent.”
The Chiral Shift Reagent (25 mL) was added to the 1H-NMR sample of the epoxy acetate product via automatic delivery pipet. A 1H-NMR spectrum of the sample was obtained. The sample was removed from the instrument and a second aliquot of Chiral Shift Reagent (25 mL) was added to the sample via automatic delivery pipet. A 1H-NMR spectrum of the sample was obtained once again. This process was repeated until the enantiomeric protons signal at 3.0 ppm shifted to approximately 3.8 ppm and split into two separate peaks. The region containing the two peaks was then expanded and the peaks were integrated in order to determine the enantiomeric ratio. A total of 125 mL of Chiral Shift Reagent was added.
1H-NMR spectra of geraniol and the epoxy alcohol as well as IR spectra of the epoxy alcohol and epoxy acetate products were obtained. A 13C-NMR spectrum of geraniol and proton decoupling NMR spectra of the epoxy acetate were provided by the instructor.
See attached page for calculations of mmol used, theoretical yield, and percent recovery from the column chromatography. See attached page for TLC plates and Rf value calculations as well.
Results
Table 1: 1H-NMR (CDCl3) Key Peak Table for Geraniol
|
Chemical Shift (ppm) |
Type of Signal |
J Values (Hz) |
Integration |
Type of Proton |
|
2.04 |
m |
—- |
21.84 |
CH2 |
|
5.07 |
d of d |
—- |
4.44 |
C=CH |
|
5.39 |
t |
—- |
4.72 |
C=CH |
*J Values were not required for this spectrum
Table 2: 13C-NMR (CDCl3) for Geraniol
|
Chemical Shift (ppm) |
Type of Carbon |
|
|
16.440 |
CH3 |
|
|
17.853 |
CH3 |
|
|
25.836 |
CH3 |
|
|
26.580 |
CH2 |
|
|
39.722 |
CH2 |
|
|
59.595 |
CH2 |
|
|
123.533 |
C=C |
|
|
124.084 |
C=C |
|
|
131.915 |
C=C |
|
|
139.986 |
C=C |
|
Table 3: Infrared Spectroscopy Peak Table for Geraniol
|
Absorption (cm-1) |
Functional Group |
|
3325.04 |
Alcohol O-H Stretch |
|
2967.67, 2916.05 |
Alkane C-H Stretch |
|
1669.10 |
C=C Stretch |
Table 4: 1H-NMR (CDCl3) Key Peak Table for Epoxy Geraniol
|
Chemical Shift (ppm) |
Type of Signal |
J Values (Hz) |
Integration |
Type of Proton |
|
3.64 |
d of d |
12.1, 6.7 |
1 |
CH2 |
|
3.76 |
d of d |
12.1, 4.1 |
1 |
CH2 |
|
5.08 |
t |
—- |
1 |
C=CH |
*J Values acquired from Reference 1
Table 5: Infrared Spectroscopy Peak Table for Epoxy Geraniol
|
Absorption (cm-1) |
Functional Group |
|
3325.04 |
Alcohol O-H Stretch |
|
2967.67, 2916.05 |
Alkane C-H Stretch |
|
1669.46 |
C=C Stretch |
|
1114.42, 1032.20 |
C—O Stretch |
Table 6: 1H-NMR (CDCl3) Key Peak Table for Epoxy Acetate
|
Chemical Shift (ppm) |
Type of Signal |
J Values (Hz) |
Integration |
Type of Proton |
|
1.33 |
s |
—- |
3 |
CH3 |
|
1.49 |
m |
—- |
2 |
CH2 |
|
1.65 |
s |
—- |
3 |
CH3 |
|
1.74 |
s |
—- |
3 |
CH3 |
|
2.08 |
t |
—- |
2 |
CH2 |
|
2.10 |
s |
—- |
3 |
CH3 |
|
3.00 |
d of d |
7.1, 4.1 |
1 |
C=CH |
|
4.03 |
d of d |
12.2, 4.1 |
2 |
CH2 |
|
4.34 |
d of d |
12.2, 7.1 |
2 |
CH2 |
|
5.08 |
t |
—- |
1 |
C=CH |
*Integration values estimated from decoupling experiment
**J Values acquired from Reference 1
Table 7: Infrared Spectroscopy Peak Table for Epoxy Acetate
|
Absorption (cm-1) |
Functional Group |
|
2970.89 2918.21 |
Alkane C-H Stretch |
|
1743.83 |
C=O Stretch |
|
1035.12 |
C—O Stretch |
Table 8: Chiral Shift 1H-NMR (CDCl3) Experiment Data
|
Sample (mg) |
Total Chiral Shift Reagent (mL) |
Chemical Shift (ppm) |
Enantiomeric Ratio |
|
5.00 |
0 |
3.00 |
—- |
|
5.00 |
25 |
3.19 |
—- |
|
5.00 |
50 |
3.35 |
—- |
|
5.00 |
75 |
3.40 |
—- |
|
5.00 |
100 |
3.50 |
—- |
|
5.00 |
125 |
3.59 |
7.39:1 |
*Chiral Shift Reagent: Eu(hfpc)3
Table 9: Class Data and Literature Values
|
Experiment |
Enantiomeric Excess |
|
Sharpless Asymmetric Epoxidation #1 |
83.5% |
|
Sharpless Asymmetric Epoxidation #2 |
76.2% |
|
mCPBA #1 |
1.6% |
|
mCPBA #2 |
4.0% |
|
J. Chem. Ed. mCPBA |
4.0% |
|
J. Chem. Ed. Sharpless Asymmetric Epoxidation |
82.0% |
Group Assigned: Sharpless Asymmetric Epoxidation #2
Table 10: Rf Values for Thin Layer Chromatography in 70% PE/30% EA
|
Product |
Rf Value |
|
Geraniol |
0.83 |
|
2,3-Epoxy Geraniol (Fractions 5-7) |
0.64 |
|
2,3-Epoxy Acetate (Fractions 6-10) |
0.95 |
*TLC Plate: Silica gel on plastic backing, 70% PE/ 30% EA, KMnO4 dip used to visualize
Due to the final product being a liquid, no melting point measurement was acquired.
Discussion
Examining Table 9: Class Data and Literature Values, it is clear that the group’s results (Sharpless Group #2) are consistent with the J. Chem. Ed. paper results. The Sharpless Asymmetric Epoxidation in both this experiment and the J. Chem. Ed. experiment demonstrated enantioselectivity. The enantiomeric excess was determined to be 76.2%, whereas the J. Chem. Ed. paper yielded 82.0% enantiomeric excess.1 Although the specific enantiomer in excess was not determined in this experiment, literature precedence demonstrates that the L-(+)-DET facilitates a Re face attack of the oxygen to the position 2,3 alkene, ultimately forming the (2S, 3S)-epoxy geraniol in excess.1,4,5
Being that geraniol contains two double bonds that are capable of reacting, there are numerous epoxide products that can form (See “Figure 2”). The mCPBA epoxidation procedure involves the addition of the peroxycarboxylic acid oxygen bound to the hydrogen adding across the double bond in a concerted mechanism (See “Figure 3”). The results indicate that there was no selectivity between the two double bonds in the mCPBA epoxidation procedure; formation of 2,3-epoxy geraniol, 6,7-epoxy geraniol, and trace amounts of 2,3,6,7-diepoxy geraniol were observed. Thus, it can be concluded that the mCPBA epoxidation procedure does not exhibit regioselectivity or stereoselectivity; the peroxide can add to either face of either double bond, creating a racemic mixture. On the contrary, the Sharpless Asymmetric Epoxidation procedure selected the double bond at the 2,3 alkene position. The Sharpless Asymmetric Epoxidation mechanism (see “Introduction”) involves the coordination of the Ti(OiPr)4 to the allylic alcohol and L-(+)-DET; the oxygen of the allylic alcohol nucleophilically attacks the titanium atom to form the complex. This coordination places the position 2,3 alkene in close proximity to the Ti(OiPr)4/L-(+)-DET complex, and ultimately, the complex has a “directing effect” that facilitates the delivery of the peroxide oxygen to the bottom face of the position 2,3 alkene. Thus, the Sharpless Asymmetric Epoxidation procedure exhibits regioselectivity and stereoselectivity. The results support this idea, as the (2S, 3S)-epoxy alcohol was the major isomer for the Sharpless Asymmetric Epoxidation.
Column chromatography was utilized for the isolation and purification of the desired epoxy geraniol and epoxy acetate (see “Introduction”). The crude epoxy geraniol product for the mCPBA epoxidation procedure contained a mixture of the two monoepoxide products and the diepoxide product; the Sharpless Asymmetric epoxidation was selective to the 2,3-epoxy geraniol, but column chromatography allowed any of the 6,7-epoxy geraniol and 2,3,6,7-diepoxy geraniol to be isolated from the desired 2,3-epoxy geraniol. Column chromatography separated these products based on their polarity. The column was packed with silica gel dissolved in a specific eluent; the eluent used for the separation of the epoxy geraniol was 70% petroleum ether and 30% ethyl acetate. Silica gel is polar and thus, the more polar products interact with the column and elute slower. The crude product is loaded onto the silica gel column, packed down with a thin layer of sand, and the eluent is forced via air pressure through the column. The products elute in order of least polar to most polar: 2,3-epoxy geraniol, then 6,7-epoxy geraniol, and lastly, 2,3,6,7-diepoxy geraniol.1
Fractions are collected in 5 mL portions, and thin-layer chromatography was utilized to determine which fractions contained the various products. Each fraction was spotted on the TLC plate and the same solvent system utilized in the column was utilized for TLC. Each epoxide product exhibited a specific Rf value, and therefore, the fractions containing the different products could be isolated and concentrated to dryness to yield the desired purified epoxy alcohol.1 The desired epoxy geraniol was then used for acetate formation step of the experiment. The both monoepoxide products were isolated for the mCPBA reaction, and only the 2,3-epoxy geraniol was isolated for the Sharpless Asymmetric Epoxidation. Column chromatography was utilized to separate the desired epoxy acetate product using the same principles outlined above. However, the solvent system used for the column and TLC was changed from 70% petroleum ether and 30% ethyl acetate to 95% petroleum ether and 5% ethyl acetate. The epoxy acetate is less polar than the epoxy geraniol, and thus, a less polar solvent system allows for a better separation of the epoxy acetates.
Once the desired epoxy acetate product was isolated, the enantiomeric ratio of each product was determined using chiral shift 1H-NMR spectroscopy. Enantiomers cannot be distinguished via 1H-NMR due to the achiral environment, and thus, a chiral shift reagent, Eu(hfpc)3, was added to the NMR sample to create a chiral environment.6 The Eu3+ acts as a Lewis Acid coordinates to the carbonyl oxygen of the acetate, and induces a downfield shift in the signal generated by the epoxide proton. The chiral lanthanide shift reagent induces this downfield shift due to its unpaired electrons in its f orbital, ultimately causes a deshielding affect on the epoxide proton.8 The chiral shift reagent was added in aliquots and the 1H-NMR spectrum was examined after each aliquot until the signal shifted significantly downfield and split into two peaks. Each peak represents each enantiomer of the product, and the enantiomeric ratio was determined using the integration values for each peak. The larger integration value was divided by the smaller integration value to yield the enantiomeric ratio.1,2 The enantiomeric excess was then easily determined from these integration values by finding the sum of the integration values and determining the percentage of each enantiomer present. Subtracting the smaller percentage value from the larger percentage value yielded the enantiomeric excess. Although the experiment did not involve the determination of which peak corresponded to each specific enantiomer, literature precedence asserts that the mCPBA epoxidation procedure did not favor a specific enantiomer, whereas the Sharpless Asymmetric Epoxidation favored the (2S, 3S)-epoxy geraniol isomer.1
As shown in Table 9 above, the mCPBA showed minimal enantioselectivity; the enantiomeric excess was 1.6% and 4.0% for the two groups performing the procedure. However, the Sharpless Asymmetric Epoxidation showed a significant amount of enantioselectivity, yielding 83.5% and 76.2% enantiomeric excess values. The J. Chem. Ed. values were 4.0% and 82.0% enantiomeric excess for the mCPBA epoxidation procedure and the Sharpless Asymmetric Epoxidation, respectively. Therefore, the class data values are in agreement with the J. Chem. Ed. article values; the mCPBA epoxidation did not show enantioselectivity, whereas the Sharpless Asymmetric Epoxidation showed significant enantioselectivity for the (2S, 3S)-epoxy geraniol isomer.
Conclusion
The purpose of this experiment was to investigate the enantioselectivity of two epoxidation procedures.1,2 The epoxidation of geraniol using the mCPBA epoxidation procedure and the Sharpless Asymmetric Epoxidation was studied in detail. After extensive analysis of chiral shift 1H-NMR spectra, it is clear that the mCPBA epoxidation procedure did not show enantioselectivity. Both the 2,3-epoxy geraniol and the 6,7-epoxy geraniol formed, and the enantiomeric excess was determined to be 1.6% and 4.0% in mCPBA groups 1 and 2, respectively. The mCPBA attacked both double bonds and from both the top and bottom face. A minimal amount of the diepoxide product formed. Thus, the hypothesis was correct.
After extensive analysis of chiral shift 1H-NMR spectra, it is clear that the Sharpless Asymmetric Epoxidation procedure favored one enantiomer of the 2,3-epoxy geraniol in 83.5% and 76.2% enantiomeric excess. The TBHP oxygen attacked the Re face of the geraniol, forming the 2S, 3S isomer in exxess. Thus, the hypothesis was correct.
Future work could include 13C-NMR spectroscopy to further characterize the product, as well as a COSY experiment to further analyze the 1H-NMR spectra.
References
- Bradley, L.; Springer, J.; Delate, G.; Goodman, A. “Epoxidation of Geraniol: An Advanced Organic Experiment that Illustrates Asymmetric Synthesis,” J. Chem. Ed. 1997, 1336-1338.
- “Determining Enantiomeric Ratios For The Epoxidation Products of Geraniol”, Dr. Lynn Bradley, The College of New jersey.
- Brown, William, Christopher Foote, Brent Iverson, and Eric Anslyn. Organic Chemistry. 5th ed. Brooks/Cole, 2008. Print.
- “Lecture Notes on Sharpless Asymmetric Epoxidation,” Dr. Lynn Bradley, The College of New Jersey.
- Ahlberg, P. “Advanced Information on the Nobel Prize in Chemistry 2001,” The Royal Swedish Academy of the Sciences. 2001, 1-12.
- “NMR Chiral Shift Reagent Experiment on the Epoxy Acetates – Determining the Enantiomeric Ratios,” Dr. Lynn Bradley, The College of New Jersey.
- “Evaluation of ee by chiral GC and by 1H-NMR with the chiral shift reagent Eu(hfc)3,” Dr. Mircea D. Gheorghiu, Massachusetts Institute of Technology.
- “Lanthanide Induced Shifts,” Dr. Hans. J. Reich, University of Wisconsin.