Determining the Effect of Acetaminophen on Peroxidases in Pisum sativum var. macrocarpon by Altering Acetaminophen Concentrations During Watering
By: Marisa Peine and Abigail Lukowicz
Abstract
Increasing amounts of pharmaceutical medications have been found in water treatment plants, leading to a concern for the ecological implications of these drugs. Research suggests that the effect of acetaminophen, particularly, on humans and mice is dependent on the levels of GSH peroxidase. If the levels are too low to control the acetaminophen, the drug becomes toxic to the organism, including plants, as many studies suggest, whose detoxification mechanisms are similar to the mammalian system. On this basis, we tested and observed the effects of acetaminophen on sugar snap peas, and questioned whether peroxidase activity levels would increase when acetaminophen was introduced into the plants’ water supply. We watered three different plants with differing concentrations of acetaminophen and found that, as the amount of acetaminophen present rose, the amount of peroxidase activity rose correspondingly. Thus, we accepted our hypothesis and concluded that, similar to humans and animals, acetaminophen toxicity in plants is dependent on the ability of the peroxidases to detoxify.
Introduction
Recently, an increasing amount of pharmaceutical medications have shown up in water treatment plants, causing a heightened interest in the effects of these pharmaceutical medications on the ecological system, especially water quality and the surrounding plant and animal life (Nikolaou, 2006). More specifically, recent studies have found higher and higher concentrations of acetaminophen (also called Paracetamol) in drinking water (Stackelberg et all, 2004). Through research, we know that when large concentrations of acetaminophen are introduced to the body, it must be detoxified by glutathione (GSH). If it is not, the acetaminophen covalently bonds to the cysteine groups of the proteins, proving to be toxic to the organism (James et all, 2003). When the effect of acetaminophen on mice was studied, it was found that a normally fatal dose of acetaminophen could be tolerated if the mice were given an intravenous injection of the glutathione peroxidase (Mirochnitchenko et all, 1999). Several subsequent research studies have concluded that acetaminophen toxicity is influenced by the level of GSH peroxidase (James, 2003; Mirochnitchenko et all, 1999).
A plant’s mechanism for detoxification is very similar to that of the mammalian system, which is also dependent on the glutathione peroxidase (Huber et all, 2008). Recent research has suggested that plants are able to take up and detoxify the acetaminophen, pointing to their great potential in water treatment (Huber et all, 1999). Many questions remain, though, before scientists are ready to use plants in this capacity. There is yet no data on the impact of long-term exposure to the acetaminophen, as well as the effect of varied concentrations of the acetaminophen on the plant. In our experiment, we wanted to look at the effect of acetaminophen, in varied concentrations, on the activity of the peroxidases within a sugar snap pea plant.
Our hypothesis is that if we water a sugar snap pea plant with an acetaminophen water solution, we will see an increase in peroxidase activity within the plant. This increase will occur because as the amount of acetaminophen within a plant rises, a greater amount of active peroxidase will be required to prevent this acetaminophen from bonding to the cysteine groups of the protein and becoming fatal to the plant.
To test this hypothesis, we watered three different sugar snap pea plants with 45 mL of either just distilled water, 1.2 mg/mL, or a 12 mg/mL acetaminophen concentration three times over the course of a week. At the end of the week, we observed the plants’ physical appearance, as well as tested the peroxidase activity and amount through a gel electrophoresis test. The plants that were receiving the acetaminophen solutions began to show signs of death in their leaves and stems, while the control plant that received strictly water showed now abnormal signs of death and appeared very healthy.
Methods
For our general experimental strategy, we used three small Pisum sativum var. macrocarpum, or sugar snap pea plants. The plants were each watered three times over the course of a week. Our control plant received 45 mL of regular water each time, the second plant was watered with 45 mL of 1.2mg/mL-dissolved acetaminophen in distilled water, and the third plant was given 45 mL of 12 mg/mL dissolved acetaminophen in distilled water. After each of these treatments, we initially observed the external effect of the acetaminophen on the plants, but in order to determine the change in peroxidase concentration within each of our plants, we used a process known as gel electrophoresis.
For each of the three plants, we took .5g samples of the roots and stem, cut the plant tissue into small pieces, and then added .5 mL of 1X Working Extraction Buffer Solution (0.3 mM NaCl, 0.03 mM Mg2Cl, 0.0002% Np-40, and 0.20 mM Tris-HCL, pH 8.0; stored at 4° C) and ground the samples into a paste with a mortar and pestle. We then transferred even amounts of the samples to 1.5mL microcentrifuge tubes and put them into the microcentrifuge to separate the supernatant from the plant tissue. We then used micropipettes to obtain 15 ul of each extract. This 15 ul was combined with 15 ul of Sample Loading Buffer (100 mM Tris, 770 mM glycine, pH 8.6, 40% glycerol, 0.04% bromophenol blue, stored at 4° C) and stored on ice until it was loaded into the electrophoresis gel wells.
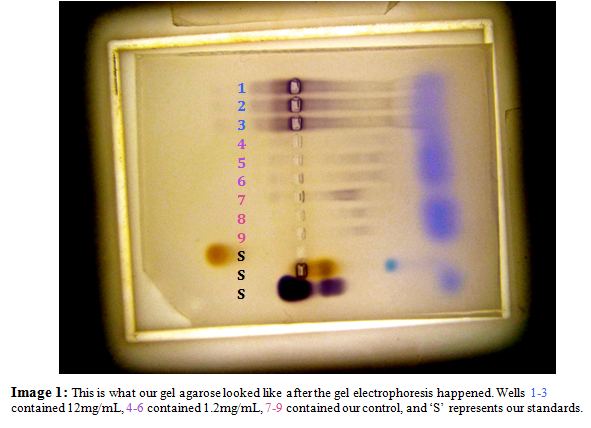
The agarose gel used for the gel electrophoresis was prepared by first combining agarose gel powder with 40 mL of 10X Tris-Glycine Stock (25 mM Tris, 192 mMglycine, pH 8.6). We then microwaved the mixture in order to allow the agarose to go into solution. The final solution was then poured into an electrophoresis gel bed, where a twelve well, plastic comb was inserted in the center of the gel and the entire bed was allowed to solidify for twenty minutes. When the agarose gel had finally solidified, we filled the gel electrophoresis box with Electrophoresis Running Buffer (1X Tris-Glycine) and then loaded the gel with 15 ul of three standards, HPR mixture, Hemoglobin and Serum Albumin, and Cytochrome C, as well as 15 ul from each of our samples. Once all of the wells had been filled, we then ran the electrophoresis machine at 150 volts for about twenty to thirty minutes.
When the gel electrophoresis machine was finished, we put the gel in an orbital shaker with Working Stain Solution (16.8 Tris-HCL, 0.0832 mg/mL chloronapthol in methanol, 0.108% H2O2, stored at 4° C) in order to develop the gel. We took two photographs of the gel, one with a clear, graph-like covering that allows us to count how far the peroxidases have traveled, and one without. These pictures allowed us to determine how many peroxidases were present as well as the activity and amount of peroxidase in each treatments and standards.
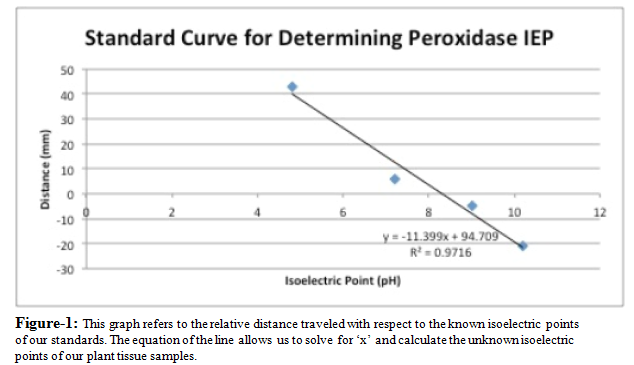
Using the photograph of the gel, the first thing that we were able to do was observe the different peroxidases present in each sample based on the stained bands that showed up on the gel. We then measured the relative distance that these peroxidases traveled and compare this distance with a standard curve line made from the relative distance that the three standards traveled and their known isoelectric points in order to find the isoelectric points for each of the peroxidase bands. Both the relative distance that the peroxidase bands travels and the isoelectric points of each of these peroxidases allow us to compare the active peroxidases in each sample. Not only could we determine how many different peroxidases were present in each sample, but we were also able to compare the peroxidases from the control group tell which told us which peroxidases were present in multiple samples.
The second thing that we did with the photograph of the gel was import it into Image J, a computer software that allows us to graph the relative density of the pixels in each of the bands on the agarose gel. Based on this data, we were able to determine how much of the peroxidase was present, as well as the activity of each of the peroxidases.
Results
After the experiment was completed, we observed some definite trends in the way our plants reacted to the acetaminophen. For our control plant, we watered it three times with 45 mL of distilled water over the span of a week and observed that the plant was bright green and the leaves were soft and looked healthy. For our second plant, which was watered 45 mL of 1.2mg/mL of acetaminophen in distilled water, there was some bright green coloring on the stem, but the leaves were beginning to brown and were becoming dry. Our third plant, which was given 45 mL of 12 mg/mL of acetaminophen in distilled water, was no longer a bright green color, but instead the stem was a light brownish grey color and the leaves were completely dried up. The plant’s steam was brittle and the crispy leaves fell off easily at the slightest touch. In general, it seems that as the concentrations of acetaminophen increased, the observed healthiness of our plant decreased.
When we ran the plant tissue from each of the three plants through the electrophoresis machine, we were able to find the isoelectric points of the active peroxidases in each of the tissues by comparing the distance they traveled to our standards’ activity using the equation of the line in Figure 1. Knowing these values, we found that the three plants shared one isoelectric band at 1.209. An interesting trend that was observed was that these common bands were the lightest in the control group, a mid-color for the second plant watered with the 1.2 mg/mL concentration of acetaminophen solution, and darkest for the third plant which was watered with the 12 mg/mL acetaminophen solution. In addition to these shared bands, the second plant displayed another isoelectric band further out at 7.168. The third plant’s isoelectric points mimicked the second plant, displaying the same second band at 7.168, as well as two additional bands at 9.274 and 10.151.
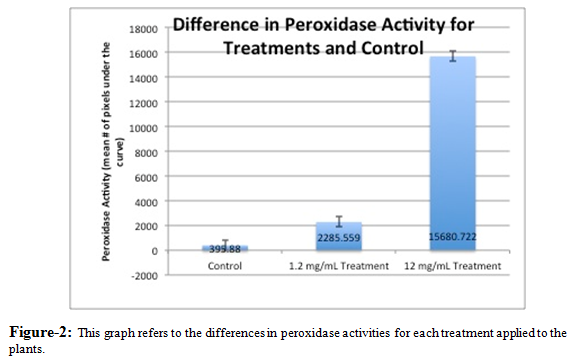
As portrayed by Figure 2, a graph of the relative density of the pixels in each isoelectric band revealed the amount and activity level of the peroxidases present in each sample. With the plant that received the 12 mg/mL acetaminophen solution, we observed an extremely high peroxidase activity, as seen in Figure 2. The plant with the 1.2 mg/mL acetaminophen solution and the control plant had significantly lower peroxidase activity.
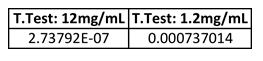
After we used Image J to find the relative density of the pixels in each of the bands on the agarose gel, we were able to quantitatively analyze our data using the numerical results we obtained. We performed a t-test for our 12mg/mL results and also for our 1.2mg/mL results. The numbers we attained for both concentrations were significantly smaller than .05, causing us to reject the null hypothesis and accept our hypothesis.
Discussion
While performing this experiment, we expected that if a sugar snap pea plant was watered with an acetaminophen water solution, we would see an increase in peroxidase activity within the plant. What we observed was that after watering individual plants with a higher and lower concentration of acetaminophen and just water, the plant given just water was healthy, bright green and had all of its leaves intact, the plant given a lower concentration of acetaminophen (1.2mg/L) was still green, but the leaves were starting to shrivel and become dry, and the plant given the highest concentration of acetaminophen (12mg/L) turned a brown color and the majority of that plant was brittle, crispy and dying.
We concluded that as one increases the concentration of acetaminophen, the plant must increase its peroxidase activity in order to attempt to keep its internal environment in equilibrium. Once the concentration becomes too toxic, the plant can no longer keep up with how much acetaminophen is present, which ultimately results in the death of the plant. An increase in peroxidase activity could happen due to how the concentration of acetaminophen and the size of the plant affects the activity of the peroxidases present within the plant, how big of a dose of acetaminophen is being administered to the plant at a given time, and how often the plant is watered with an acetaminophen solution.
While performing this experiment, questions as to “What are the effects of acetaminophen on plants?”, “What will happen to the peroxidase activity levels when acetaminophen is introduced into their water?” and the biggest question “Why do we care?” ran through our minds. Since increasing amounts of pharmaceutical medications have been found in water treatment plants, and later in the water which comes from them, this has lead to a concern about the ecological implications of these types of drugs (Nikolaou, 2006.). Research on pharmaceuticals suggests that the effect of acetaminophen, particularly, on humans and mice is largely dependent on the levels of GSH peroxidase (James, 2003). It has been found that if the peroxidase levels are too low to control the concentration of acetaminophen, the drug becomes toxic, and depending on how high a concentration, could result in death (Mirochnitchenko et all, 1999). If pharmaceutical concentrations become too high in the ground water supply, large areas of plant could be killed, posing a threat to the ecological equilibrium and habitats of insects and animals that rely on those plants to survive.
The next logical steps of our topic would be to measure the concentration of acetaminophen still in human urine when it first enters the water treatment plant, then measure the concentration after it has gone through the treatment plant. Upon finding the final concentration, one could use that concentration to make duplicate solutions and use test solutions on plants that are commonly found surrounding water treatment plants. Instead of using smaller plants, we could work with larger plants that have greater area to them. While using these larger plants, we would also test them for longer periods of time to see how the concentrations of acetaminophen found in water after being treated affects plants for extended time periods. In addition to these options, we could test plants commonly consumed by humans to see if acetaminophen and other pharmaceuticals could have a negative impact on plants we currently rely on to survive.
Recent research has suggested that plants are able to take up and detoxify the acetaminophen, pointing to their great potential in water treatment (Huber et all, 1999). A couple questions remain, though, before scientists are ready to use plants in this capacity. There is yet no data on the impact of long-term exposure to the acetaminophen, as well as the effect of varied concentrations of the acetaminophen on the plant, so further research into how pharmaceuticals affect a plant’s ability to rid itself of the toxins could become a bigger focus as the pharmaceutical industry continues to grow.
Literature Cited
Huber, Christian; Bartha Bernadett; Harpaintner, Harper; Schroder, Peter.
“Metabolism of acetaminophen (paracetamol) in plants—two independent pathways result in the formation of a glutathione and a glucose conjugate.” Environmental Science and Pollution Research. 16.2. p. 206-213. 2009. Springer. Web. 11 March. 2013. http://link.springer.com/article/10.1007%2Fs11356-008-0095-z?LI=true#
This study looked at the effect of acetaminophen on plants. They found that the plants take it up and are able to detoxify it.
James, Laura P., Philip R. Mayeux, and Jack A. Hinson. “ACETAMINOPHEN-INDUCED
HEPATOTOXICITY.” ACETAMINOPHEN-INDUCED HEPATOTOXICITY. Drug Metabolism and Disposition, Dec. 2003. Web. 07 Mar. 2013. <http://dmd.aspetjournals.org/content/31/12/1499.full>.
This article specifically focuses on the metabolic activity of acetaminophen, looking particularly at the covalent bond toxicity and the factors, such as oxidative stress, nitrotyrosine formation, etc., that influence this.
McNeil Consumer Healthcare (2013). A Pharmacologic Overview of Tylenol (acetaminophen). Web. 11 March 2013.
<http://www.tylenolprofessional.com/pharmacology.html>
Mirochnitchenko, Oleg, Miriam Weisbrot-Lefkowitz, Kenneth Reuhl, Chung Yang, and Masayori
Inouye. “Acetaminophen Toxicity: Opposite Effects of Two Forms of Glutathione Peroxidase.” Acetaminophen Toxicity. The Journal of Biological Chemistry, 9 Apr. 1999. Web. 08 Mar. 2013. <http://www.jbc.org/content/274/15/10349.full>.
This research paper looked at how acetaminophen specifically works in the body by studying it’s effects if taken in an overdose amount. They also tested several prevention treatments to discover more about it’s process of metabolism, going into detail about how and why certain result would occur.
Nikolaou, Anastasia, Sureyya Meric, and Despo Fatta. “Occurrence Patterns of
Pharmaceuticals in Water and Wastewater Environments – Springer.” Occurrence Patterns of Pharmaceuticals in Water and Wastewater Environments – Springer. Annual Biannual Chem 01 Feb. 2007. Web. 11 Mar. 2013. <http://link.springer.com/article/10.1007/s00216-006-1035-8?LI=true>.
This paper is a compilation of recent research into the sources, occasions, and results of pharmaceutical medications in water. Our interest was particularly related to the information it contained about the occurrence of acetaminophen in water. We view this as a possible concern because an increase in acetaminophen use leads to the presence of acetaminophen in our water systems.
Stackelberg, Paul E., Edward T. Furlong, Michael Meyer, Steven Zaugg, Alden Henderson, and
Dori Reissman. “Persistence of Pharmaceutical Compounds and Other Organic Wastewater Contaminants in the Conventional Drinking-water-treatment Plant.” DigitalCommons. University of Nebraska- Lincoln, 2004. Web. 10 Mar. 2013. <http://digitalcommons.unl.edu/usgsstaffpub/443/>.
This research studied the water that went through water-treatment plants and evaluated what was in the water, what the plant was able to remove, and what remained when the water went back into the recycle system.
